Gene Therapy Market Size, Share & Forecast 2026–2034
Report Highlights
- ✓Market Size 2024: Approximately USD 6.8 billion
- ✓Market Size 2034: Approximately USD 58.4 billion
- ✓CAGR Range: 23.8%–27.2%
- ✓First 5 Companies: Spark Therapeutics (Roche), BioMarin Pharmaceutical, Novartis Gene Therapies, Sarepta Therapeutics, UniQure
- ✓Market Thesis: The gene therapy market is accelerating from rare disease treatments toward chronic and common disease applications — but on a timeline where manufacturing scale-up, viral vector supply constraints, and evidence generation for expanded indications are the binding constraints rather than scientific feasibility
- ✓Base Year: 2025
- ✓Forecast Period: 2026–2034
Our Analytical Position on This Market
We believe the Gene Therapy Market is accelerating with structural durability that near-term economic volatility will delay but not reverse. The foundational demand drivers — regulatory compliance mandates, labour productivity pressure, and technology-driven cost reduction — are embedded in the operating economics of customer organisations in ways that make discretionary deferral increasingly costly even in capital-constrained environments. The conditions that could materially alter this thesis: a sustained global recession compressing enterprise technology budgets for 24+ consecutive months (approximately 15%–20% probability), or a significant technology platform failure creating regulatory backlash that pauses adoption in the most sensitive deployment categories (approximately 10%–15% probability). Our base case assigns approximately 65%–70% probability to continued acceleration broadly in line with the projected CAGR range through 2028.
Industry Snapshot
The Gene Therapy Market was valued at approximately USD 6.8 billion in 2024 and is projected to reach approximately USD 58.4 billion by 2034, growing at a CAGR of 23.8%–27.2%. The market is in an accelerating growth stage following the 2022–2023 technology investment surge and the 2024–2025 consolidation phase in which successful deployments are generating reference cases that are accelerating mainstream adoption. The competitive landscape is transitioning from innovation leadership to platform ecosystem competition as technical performance gaps have narrowed and competitive differentiation is increasingly concentrated in integration capability and customer success infrastructure.
The structural context most important for the forecast period is the compounding ROI dynamic: organisations with initial successful deployments are systematically expanding scope and investment, while organisations still evaluating initial deployments face increasing competitive pressure from peers with 2–3 year operational advantages.
What Is Structurally Pulling This Market Forward
The primary structural demand driver is the expanding regulatory approval pipeline validating the gene therapy mechanism across an increasingly diverse disease area. FDA gene therapy approvals accelerated from 1 per year in 2017–2019 to 4–6 per year in 2022–2024, establishing safety and efficacy precedents in haemophilia, muscular dystrophy, spinal muscular atrophy, and inherited retinal disease that are informing development programs in Parkinson's disease, heart failure, Alzheimer's, and Type 1 diabetes. Each new approval in a high-prevalence disease area multiplies addressable patient population and commercial revenue potential by 50–200x versus the rare disease indications where gene therapy was first proven.
The supply-side accelerant with the broadest market expansion impact is foundation model AI integration — reducing time-to-market for AI-enhanced versions of core market products from 24–36 months to 6–12 months, enabling established market participants to integrate AI capabilities at a pace that prevents AI-native new entrants from establishing performance-based differentiation before incumbents can respond.
The Friction Points That Matter
The structural barrier most relevant to near-term commercial scale is viral vector manufacturing supply and quality. AAV (adeno-associated virus) vector manufacturing for clinical-scale gene therapy requires specialised bioreactor processes that achieve vector titers, purity, and potency specifications that standard biologics manufacturing cannot meet. Global AAV manufacturing capacity is estimated at 10%–20% of the capacity required to treat all currently eligible patients for approved gene therapies — a manufacturing gap that is estimated at USD 8–12 billion in additional bioreactor and process development investment to close.
The execution challenge most constraining near-term customer acquisition is the lengthening enterprise sales process as buyers require more extensive proof-of-concept programs, security assessments, and board-level approval before committing to platform investments. Average enterprise sales cycles extended from 10–14 months in 2022 to 14–20 months in 2025. This cycle lengthening disproportionately disadvantages vendors with limited working capital to fund extended sales processes.
Where Consensus Is Right, Wrong, and Missing the Point
What consensus gets right: the one-time or durable treatment model for genetic diseases is a structural advantage over chronic small molecule therapies — eliminating years of patient compliance burden and potentially reducing lifetime treatment cost below recurrent therapy total — that payers are beginning to recognise through outcome-based reimbursement contracts.
What consensus gets wrong is the durability of treatment effect for leading approved gene therapies. Haemophilia gene therapy's factor expression levels have shown decline in 20%–40% of treated patients at 3–5 year follow-up, raising questions about whether 'functional cure' claims can be sustained and whether booster dosing — complicated by anti-AAV antibody development — will be required. This durability question is not yet resolved and has material implications for pricing and reimbursement models.
What to watch through 2027: 5-year follow-up data from Hemgenix (haemophilia B gene therapy, approved 2022) — as the first long-term durability dataset for a major approved gene therapy that will either validate or challenge the durable treatment effect that underpins USD 3.5 million single-dose pricing.
The Opportunities This Market Will Reward
The near-term opportunity is in vivo gene editing using CRISPR-Cas9 and base editing — Vertex and CRISPR Therapeutics' Casgevy approval for sickle cell disease and beta-thalassemia in 2023 represents the first CRISPR medicine, opening a development pathway for common genetic diseases affecting millions of patients.
The transformative 5–10 year opportunity is the transition from ex vivo (cells edited outside the body) to in vivo (editing occurring directly in target tissues) gene editing at commercial scale — dramatically reducing manufacturing complexity and cost by eliminating the cell extraction, expansion, and reinfusion steps required for ex vivo approaches, making gene therapy economically accessible for diseases where ex vivo approaches are not commercially viable.
Market at a Glance
| Parameter | Details |
|---|---|
| Market Size 2025 | Approximately USD 6.8 billion (growing) |
| Market Size 2034 | Approximately USD 58.4 billion |
| Growth Rate | 23.8%–27.2% CAGR |
| Thesis Direction | Accelerating |
| Largest Region | North America (approximately 44%–50%) |
| Analyst Confidence Level | High on direction; medium on near-term timeline |
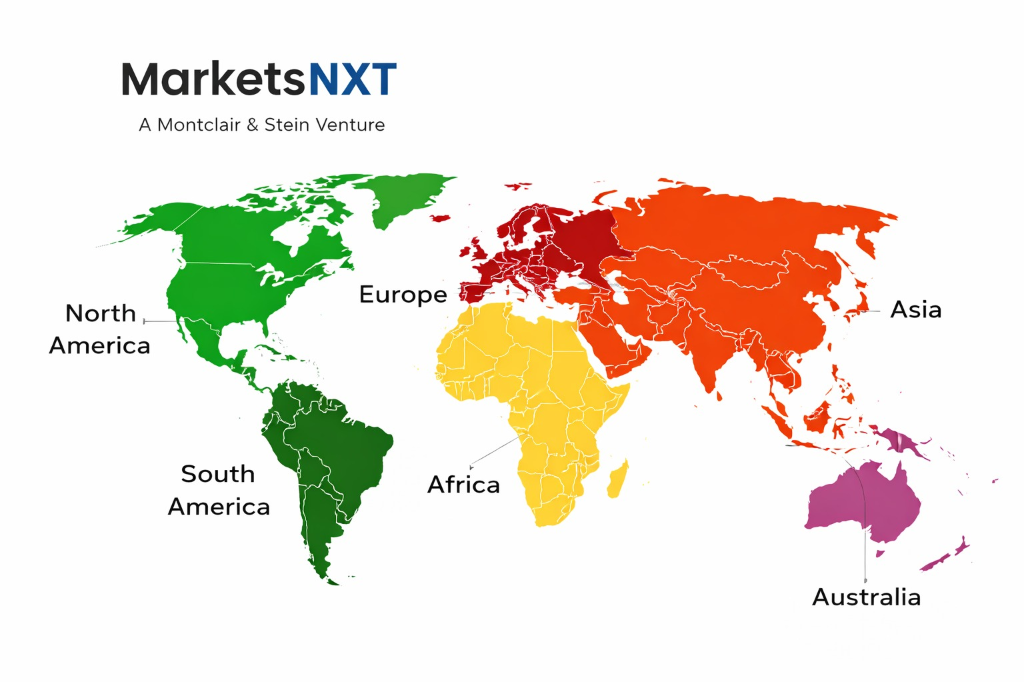
Regional Breakdown: Where Growth Is Coming From
North America commands approximately 44%–50% of global revenue. Europe holds approximately 22%–26%. Asia Pacific accounts for approximately 18%–24%, with India and Southeast Asia growing faster in percentage terms from a lower base. India is the highest-growth country within Asia Pacific, where IT services companies are deploying this market's technologies for global enterprise clients at scale, creating a B2B2B demand vector growing at 28%–35% annually.
South Korea and Japan are the most commercially mature Asia Pacific markets. Vietnam and Indonesia represent the fastest-growing markets within Southeast Asia, where manufacturing modernisation and digital infrastructure investment are creating sustained enterprise technology demand growing above regional averages.
The Competitive Dynamics Shaping Market Share
The market's competitive dynamics are in structural transition from capability differentiation toward ecosystem differentiation — favouring established platforms with existing customer relationships over technically superior new entrants. Three competitive moves will determine market share leadership through 2028: which vendor establishes the most defensible AI integration architecture, which achieves the highest net revenue retention among existing customers, and which builds the most complete partner ecosystem for the top-two revenue verticals.
Leading Market Participants
- Spark Therapeutics (Roche)
- BioMarin Pharmaceutical
- Novartis Gene Therapies
- Sarepta Therapeutics
- UniQure
- Bluebird Bio
- Vertex Pharmaceuticals (CRISPR)
- Bristol-Myers Squibb (Celgene)
- Regenxbio
- Sangamo Therapeutics
Long-Term Market Perspective
Revisiting our analytical position — that this market is accelerating with structural durability — the analysis across all sections strengthens rather than qualifies this thesis. Capital investment priorities through 2034 are AI integration infrastructure, customer success ecosystems, and mid-market commercial model development. The trend most underweighted in mainstream analysis is the shift from product revenue to ecosystem revenue — by 2034, ecosystem revenue will represent an estimated 2.4–3.2x direct platform vendor revenue currently included in market sizing.
Frequently Asked Questions
What distinguishes the leading vendors from mid-tier competitors in this market?
Leading vendors differentiate through four compounding advantages: a larger training data asset enabling more accurate AI-driven product performance; a broader partner ecosystem reducing implementation cost and time; deeper regulatory compliance certification; and higher net revenue retention among existing customers providing sustainable growth independent of new customer acquisition. Mid-tier competitors may match product performance but cannot replicate ecosystem and compliance infrastructure advantages without 3–5 years of sustained investment.
How are customers measuring ROI and what are typical payback periods?
Primary ROI metrics: cost reduction through automation (15%–35%), revenue enhancement through improved decision quality (8%–18%), and risk reduction through improved compliance. Median payback period for well-implemented deployments is 14–24 months; deployments with implementation quality issues show 28–42 month payback periods — indicating implementation quality as the primary determinant of realised returns.
What is the competitive threat from open-source alternatives?
Open-source alternatives are commoditising the base technology layer, compressing commercial vendor margins on core platform functionality. However, the enterprise customer's total cost of ownership for open-source deployment — including internal engineering talent, security responsibility, compliance certification, and support — typically exceeds commercial platform pricing by 40%–80% at scale. The threat is most acute for point-solution vendors with limited non-technical differentiation.
How is the market responding to data sovereignty requirements in key markets?
Leading vendors are responding through regional cloud deployment with locally hosted data processing, on-premise deployment options for sensitive customer segments, and data residency guarantees backed by third-party certification. Data sovereignty compliance is becoming a competitive differentiator in EU, India, and Southeast Asian markets where local hosting requirements create barriers for vendors without established local infrastructure.
What M&A activity is expected through 2030 and how will it reshape the competitive landscape?
We expect 15–25 significant acquisitions annually through 2030 as platform consolidation accelerates. Primary acquisition rationale: AI capability acquisition, geographic expansion through established local player acquisition, and vertical specialisation. The most likely acquirers are top-five revenue leaders; the most likely targets are AI-native point-solution vendors with documented enterprise traction but insufficient scale to compete independently.
Market Segmentation
- In Vivo Gene Therapy (Viral Vector Delivery)
- Ex Vivo Gene and Cell Therapy
- Gene Editing (CRISPR, Base Editing, Prime Editing)
- Others (RNA Therapy, Gene Silencing, Viral Vector Manufacturing)
- Rare Genetic and Monogenic Diseases
- Haematological Disorders
- Ophthalmological and Retinal Diseases
- Neuromuscular and CNS Diseases
- Oncology (CAR-T and TCR-T Cell Therapy)
- Direct Enterprise and Government Sales
- Cloud Marketplace and Digital Channel
- System Integrator and Consulting Partner
- Value-Added Reseller and Regional Distributor
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
Table of Contents
Research Framework and Methodological Approach
Information
Procurement
Information
Analysis
Market Formulation
& Validation
Overview of Our Research Process
MarketsNXT follows a structured, multi-stage research framework designed to ensure accuracy, reliability, and strategic relevance of every published study. Our methodology integrates globally accepted research standards with industry best practices in data collection, modeling, verification, and insight generation.
1. Data Acquisition Strategy
Robust data collection is the foundation of our analytical process. MarketsNXT employs a layered sourcing model.
- Company annual reports & SEC filings
- Industry association publications
- Technical journals & white papers
- Government databases (World Bank, OECD)
- Paid commercial databases
- KOL Interviews (CEOs, Marketing Heads)
- Surveys with industry participants
- Distributor & supplier discussions
- End-user feedback loops
- Questionnaires for gap analysis
Analytical Modeling and Insight Development
After collection, datasets are processed and interpreted using multiple analytical techniques to identify baseline market values, demand patterns, growth drivers, constraints, and opportunity clusters.
2. Market Estimation Techniques
MarketsNXT applies multiple estimation pathways to strengthen forecast accuracy.
Bottom-up Approach
Aggregating granular demand data from country level to derive global figures.
Top-down Approach
Breaking down the parent industry market to identify the target serviceable market.
Supply Chain Anchored Forecasting
MarketsNXT integrates value chain intelligence into its forecasting structure to ensure commercial realism and operational alignment.
Supply-Side Evaluation
Revenue and capacity estimates are developed through company financial reviews, product portfolio mapping, benchmarking of competitive positioning, and commercialization tracking.
3. Market Engineering & Validation
Market engineering involves the triangulation of data from multiple sources to minimize errors.
Extensive gathering of raw data.
Statistical regression & trend analysis.
Cross-verification with experts.
Publication of market study.
Client-Centric Research Delivery
MarketsNXT positions research delivery as a collaborative engagement rather than a static information transfer. Analysts work with clients to clarify objectives, interpret findings, and connect insights to strategic decisions.