Next-Generation Sequencing (NGS) Market Size, Share & Forecast 2026–2034
Report Highlights
- ✓Market Size 2024: USD 8.0 billion
- ✓Market Size 2034: USD 47.1 billion
- ✓CAGR: 20.6%
- ✓Market Definition: Massively parallel DNA/RNA sequencing platforms, reagents, and clinical services for diagnostics and precision medicine.
- ✓Leading Companies: Illumina, Thermo Fisher Scientific, Oxford Nanopore Technologies, Pacific Biosciences, MGI Tech
- ✓Base Year: 2025
- ✓Forecast Period: 2026–2034
Who Controls This Market — And Who Is Threatening That Control
Illumina controls approximately 80% of global NGS instrument revenue, built on short-read sequencing-by-synthesis chemistry that has generated the majority of the world's sequencing data since 2007. Illumina's competitive moat is not technology — PacBio and Oxford Nanopore offer superior long-read capabilities for specific applications — it is the installed base data lock-in: the world's genomic databases (gnomAD, ClinVar, COSMIC oncology database) are built on Illumina short-read data, meaning interpretation algorithms validated on Illumina data perform less reliably on other platforms' data. Illumina's NovaSeq X series, launched in 2023 at a USD 100/genome sequencing cost target, is designed to pre-empt competition by lowering the per-genome cost barrier before long-read alternatives reach comparable cost points.
Oxford Nanopore Technologies (ONT) has built the most differentiated NGS platform through miniaturisation and portability — the MinION sequencer fits in a shirt pocket, runs on a laptop, and enables sequencing in field conditions (infectious disease outbreak response, agricultural pathogen monitoring, point-of-care clinical settings) that Illumina's benchtop instruments cannot serve. ONT's competitive advantage is read length — reads of 1 million+ bases versus Illumina's 150–300 base reads — enabling direct detection of structural variants, methylation patterns, and full-length gene isoforms that short-read assembly cannot resolve. ONT's limitation is raw accuracy (Q20 for most runs versus Q30+ for Illumina), which restricts applications requiring single-base variant calling precision.
MGI Tech, a BGI Genomics subsidiary, manufactures sequencing instruments based on DNA nanoball sequencing technology that competes directly with Illumina at 30%–50% lower instrument and reagent costs. MGI's DNBSEQ-G400 and G400RS are deployed in high-volume sequencing centres in China and increasingly globally, with more than 700 instruments installed in 60+ countries by 2023. MGI's cost advantage reflects Chinese manufacturing scale, lower R&D cost amortisation, and government support — creating price competition that is compressing Illumina's reagent margins in research and population genomics applications where instrument price rather than performance is the primary purchasing criterion.
Industry Snapshot
The global NGS market generated approximately USD 8.9 billion in 2024, with oncology applications (clinical tumour sequencing, liquid biopsy, minimal residual disease monitoring) representing approximately 35%, research and academic applications approximately 30%, non-invasive prenatal testing (NIPT) approximately 15%, and infectious disease and agricultural genomics approximately 20%. Sequencing throughput costs have declined from USD 100 million per human genome (Human Genome Project, 2003) to USD 200–500 per genome in 2024 — a 200,000x reduction in 20 years — making whole genome sequencing economically accessible for population-scale clinical applications for the first time.
The clinical diagnostics segment is the highest-growth and highest-margin application, driven by oncology companion diagnostics and liquid biopsy. Foundation Medicine's FoundationOne CDx (Roche) and Guardant Health's Guardant360 are FDA-cleared liquid biopsy and tissue NGS panel tests with companion diagnostic indications across 40+ approved therapies, creating a reimbursed clinical market where Illumina's NovaSeq X runs for USD 1,500–3,500 per patient test. The NIPT market — non-invasive prenatal chromosomal aneuploidy detection from maternal blood — represents a USD 1.2 billion global market concentrated in China (BGI's NIFTY test) and the US (Natera Panorama, Illumina-affiliated Verinata), with universal NIPT reimbursement in several European markets.
The Forces Accelerating Demand Right Now
Liquid biopsy — detecting circulating tumour DNA (ctDNA) in blood samples — is transitioning from an academic research tool to a standard-of-care oncology procedure across three clinical use cases: late-stage treatment selection (identifying actionable mutations for targeted therapy), minimal residual disease (MRD) monitoring after treatment to detect early recurrence, and multi-cancer early detection (MCED) as a cancer screening tool. Guardant Health's Guardant360 CDx and Foundation Medicine's FoundationOne Liquid CDx are FDA-approved liquid biopsy tests for treatment selection with reimbursement pathways established in the US and several European markets. GRAIL's Galleri multi-cancer early detection test, targeting 50 cancer types from a single blood draw, represents the largest potential new clinical NGS market — a USD 10+ billion annually addressable MCED market if cancer screening guidelines are updated to include liquid biopsy.
The UK Biobank (500,000 whole genomes sequenced), UK 100,000 Genomes Project (completed), All of Us Research Programme (US, targeting 1 million diverse genomes), Saudi Arabia's Saudi Human Genome Programme, and China's Precision Medicine Initiative collectively represent government-funded whole genome sequencing programmes sequencing millions of genomes annually. These programmes create concentrated, high-volume sequencing demand that drives instrument utilisation at partnered sequencing centres, providing Illumina, MGI, and ONT with high-volume reference deployments. The genomic data generated creates population-scale reference databases that improve clinical variant interpretation accuracy — a data network effect that benefits all NGS application developers.
What Is Holding This Market Back
Generating sequencing data is technically straightforward with modern instruments; interpreting the data is the bottleneck. A whole genome sequencing run produces 90–150 GB of raw data requiring alignment to the reference genome, variant calling, annotation, and clinical interpretation — a bioinformatics workflow requiring UNIX command line proficiency, genomics pipeline expertise, and clinical genetics knowledge that most hospital laboratories lack. The Association for Molecular Pathology estimates that fewer than 5,000 clinical molecular geneticists are practising in the US — insufficient for the clinical NGS scale anticipated by 2030 oncology and rare disease guidelines. Cloud bioinformatics platforms (DNAnexus, Seven Bridges, AWS HealthOmics) are automating pipeline execution, but clinical variant interpretation (distinguishing pathogenic from benign variants, identifying novel actionable mutations) requires expert human review that cannot be fully automated at current AI capability levels.
Clinical NGS reimbursement in the US is advancing through FDA companion diagnostic clearances and Medicare Coverage with Evidence Development (CED) pathways, but remains inconsistent: Foundation Medicine's FoundationOne CDx has CMS reimbursement at USD 3,500 per test for advanced solid tumour patients, but most hospital-developed NGS panels receive USD 600–1,200 per test under limited coverage, inadequate to recover sequencing and interpretation costs. In Europe, national health system reimbursement for NGS diagnostics varies from comprehensive coverage (France's INCa programme, UK's NHSE Genomics Medicine Service) to no systematic coverage in lower-income EU member states, fragmenting the commercial market and creating access inequities.
The Investment Case: Bull, Bear, and What Decides It
The bull case is CMS issuing a positive National Coverage Determination for GRAIL's Galleri multi-cancer early detection test (or an equivalent MCED product) before 2027, triggering Medicare reimbursement at USD 900–1,400 per annual test for the 65+ population. With 58 million Medicare beneficiaries and a 30%–40% estimated uptake for cancer screening tests among high-adherence populations, this creates an annual MCED test market of USD 15–25 billion — the largest single clinical test in US history. The NGS instrument and reagent demand to support this volume would require significant capacity expansion across all sequencing platforms. Bull case probability: 25%.
The bear case is CMS declining to issue a positive NCD for MCED tests pending additional randomised controlled trial evidence of mortality reduction — a standard evidence requirement that GRAIL's CCGA and PATHFINDER studies (case-ascertained, no mortality endpoint) do not meet. Without Medicare coverage, MCED market development relies on direct-to-consumer and commercial insurance uptake at USD 500–1,400 per test — a market growing but constrained to premium segments. Liquid biopsy growth continues in oncology treatment selection (already reimbursed) but the MCED mass-market inflection does not occur before 2030. Bear case probability: 45%.
The decisive data point is the GRAIL ECLIPSE trial interim results (a randomised trial with cancer detection as primary endpoint, designed to meet CMS evidence standards) expected in 2025–2026, and CMS's subsequent NCD timeline. A positive ECLIPSE result meeting CMS's sensitivity and specificity thresholds triggers the coverage decision process; a negative or inconclusive result extends the clinical validation timeline by 3–5 years. Secondary indicator: whether UnitedHealthcare, Cigna, or Aetna issue positive coverage policies for Galleri before any Medicare decision, which would indicate commercial insurer risk tolerance.
Where the Next USD Billion Is Being Built
The 3–5 year opportunity is NGS-based infectious disease surveillance and antimicrobial resistance (AMR) monitoring at hospital and public health laboratory scale. Whole genome sequencing of bacterial pathogens provides outbreak cluster analysis (identifying hospital transmission chains), resistance gene identification (guiding antibiotic therapy in 24–48 hours versus 5–7 days for culture-based methods), and real-time AMR prevalence monitoring for public health decision-making. The COVID-19 pandemic demonstrated the operational value of rapid whole genome sequencing for variant surveillance (UK's COG-UK consortium sequenced 40%+ of UK COVID cases at peak), creating a template for permanent pathogen surveillance programmes. The WHO's global AMR surveillance programme and US CDC's advanced molecular detection initiative are funding NGS pathogen surveillance infrastructure in 50+ countries.
The 5–10 year opportunity is single-cell sequencing at clinical scale for immune profiling and personalised medicine. Single-cell RNA sequencing (scRNA-seq) characterises the gene expression profile of individual cells within a tumour or immune sample, revealing the heterogeneity — the mix of cell types and their functional states — that bulk sequencing obscures. Clinical applications include: tumour immune microenvironment profiling to predict immunotherapy response, minimal residual disease characterisation at single-cell resolution, and personalised CAR-T cell therapy manufacturing optimisation. 10x Genomics (Chromium platform), Illumina (acquisition attempt of Pacific Biosciences failed), and Parse Biosciences are building the commercial platform, but clinical workflow integration and per-cell cost reduction to USD 0.10–0.50 per cell are required to enable routine clinical use by 2030.
Market at a Glance
| Parameter | Details |
|---|---|
| Market Size 2024 | USD 8.0 billion |
| Market Size 2034 | USD 47.1 billion |
| Growth Rate | 20.6% CAGR (2026–2034) |
| Most Critical Decision Factor | Technology maturity and enterprise deployment readiness |
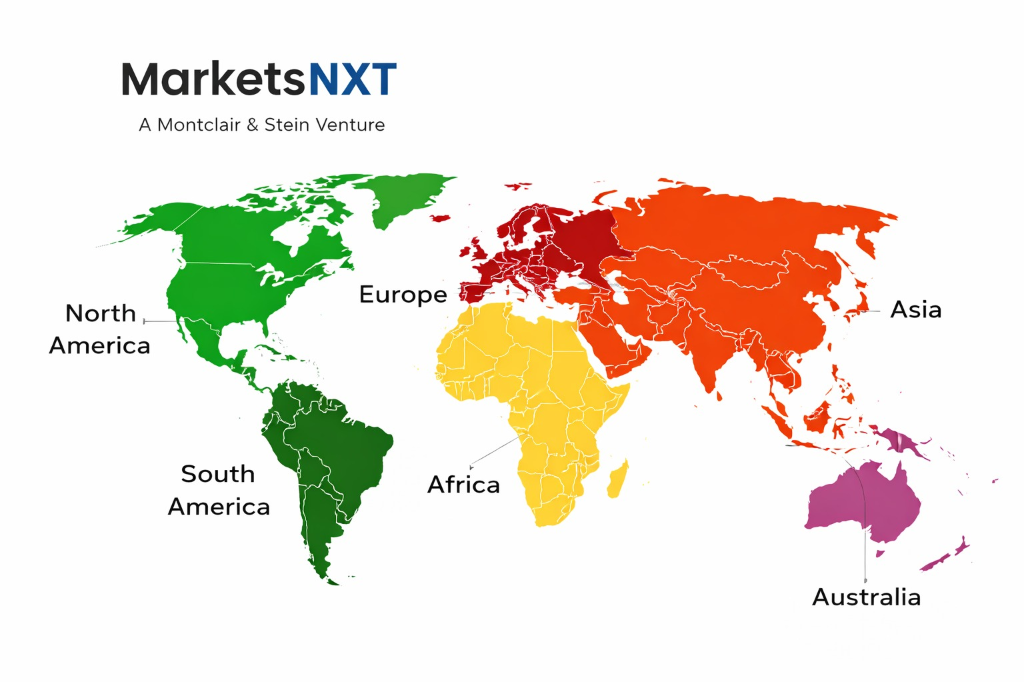
| Largest Region | North America |
| Competitive Structure | High — Illumina dominance challenged by long-read platforms and Chinese cost |
Regional Intelligence
The FDA's Center for Devices and Radiological Health (CDRH) regulates NGS-based in vitro diagnostics as Class II or Class III devices under 21 CFR Part 866, with companion diagnostics approved under Premarket Approval (PMA). The FDA's framework for Oncology NGS-Based Companion Diagnostics (published 2017) established the regulatory pathway for laboratory-developed tests and commercially distributed NGS panels, requiring analytical validation, clinical validation, and variant database submissions. The FDA's Breakthrough Device designation has been applied to multiple liquid biopsy and MCED tests, accelerating review timelines — the first MCED Breakthrough Designation was granted to GRAIL's Galleri in 2020.
The EU In Vitro Diagnostic Regulation (IVDR, fully applicable May 2022) reclassifies most clinical NGS diagnostics as Class C or Class D devices (up from Class B under the previous IVDD), requiring notified body certification, performance evaluation studies, and post-market surveillance programmes. The IVDR transition has created a certification backlog — insufficient notified body capacity to process applications from all NGS diagnostic developers simultaneously — causing launch delays of 18–36 months for new clinical NGS tests in the EU market. The EU's Cancer Mission and the European Genomic Data Infrastructure (GDI) project represent the public health investment in genomic data standardisation and cross-border data sharing that underpins European clinical NGS scale.
Leading Market Participants
- Illumina
- Thermo Fisher Scientific
- Oxford Nanopore Technologies
- Pacific Biosciences
- MGI Tech
- Agilent Technologies
- Twist Bioscience
- Guardant Health
- Foundation Medicine
- Natera
Long-Term Market Perspective
By 2034, whole genome sequencing will be a routine component of oncology care in high-income countries — as standard as CT imaging in cancer staging workflows. Liquid biopsy will be reimbursed for treatment selection, MRD monitoring, and (pending evidence accumulation) multi-cancer early detection across major Western health systems. The sequencing instrument market will have consolidated around Illumina for short-read clinical applications and Oxford Nanopore for long-read and point-of-care applications, with MGI Tech as the cost-competitive challenger in high-volume research and public health genomics. Per-genome sequencing cost will approach USD 50–100 — below the threshold at which whole-genome sequencing is economically justifiable for newborn screening programmes.
The most consequential long-term development is the integration of polygenic risk scores (PRS) into preventive healthcare. PRS — derived from NGS-analysed genetic variants associated with disease risk — can identify individuals at 3–5x population average risk of cardiovascular disease, type 2 diabetes, breast cancer, and other common conditions before any clinical symptoms appear. Population-scale PRS screening could identify 15%–20% of adults with actionable elevated risk warranting intensified surveillance or preventive intervention — transforming NGS from a diagnostic tool to a preventive medicine infrastructure. The NHS's Genomics England is the most advanced national programme implementing PRS into clinical pathways, with a target of PRS integration into primary care by 2027.
Frequently Asked Questions
Market Segmentation
- Short-Read Sequencing by Synthesis
- Long-Read Nanopore Sequencing
- Long-Read SMRT Sequencing
- DNA Nanoball Sequencing
- Emerging Single-Molecule and Spatial Transcriptomics
- Oncology
- Liquid Biopsy
- Rare and Inherited Disease Whole Genome and Exome Diagnosis
- Non-Invasive Prenatal Testing
- Infectious Disease Surveillance and AMR Monitoring
- Research Genomics
- Hospital Molecular Diagnostics Laboratories
- Pharmaceutical and Biotechnology Drug Discovery
- Academic and Government Research Institutions
- Contract Genomics Sequencing Laboratories
- Agricultural and Environmental Genomics
Table of Contents
Research Framework and Methodological Approach
Information
Procurement
Information
Analysis
Market Formulation
& Validation
Overview of Our Research Process
MarketsNXT follows a structured, multi-stage research framework designed to ensure accuracy, reliability, and strategic relevance of every published study. Our methodology integrates globally accepted research standards with industry best practices in data collection, modeling, verification, and insight generation.
1. Data Acquisition Strategy
Robust data collection is the foundation of our analytical process. MarketsNXT employs a layered sourcing model.
- Company annual reports & SEC filings
- Industry association publications
- Technical journals & white papers
- Government databases (World Bank, OECD)
- Paid commercial databases
- KOL Interviews (CEOs, Marketing Heads)
- Surveys with industry participants
- Distributor & supplier discussions
- End-user feedback loops
- Questionnaires for gap analysis
Analytical Modeling and Insight Development
After collection, datasets are processed and interpreted using multiple analytical techniques to identify baseline market values, demand patterns, growth drivers, constraints, and opportunity clusters.
2. Market Estimation Techniques
MarketsNXT applies multiple estimation pathways to strengthen forecast accuracy.
Bottom-up Approach
Aggregating granular demand data from country level to derive global figures.
Top-down Approach
Breaking down the parent industry market to identify the target serviceable market.
Supply Chain Anchored Forecasting
MarketsNXT integrates value chain intelligence into its forecasting structure to ensure commercial realism and operational alignment.
Supply-Side Evaluation
Revenue and capacity estimates are developed through company financial reviews, product portfolio mapping, benchmarking of competitive positioning, and commercialization tracking.
3. Market Engineering & Validation
Market engineering involves the triangulation of data from multiple sources to minimize errors.
Extensive gathering of raw data.
Statistical regression & trend analysis.
Cross-verification with experts.
Publication of market study.
Client-Centric Research Delivery
MarketsNXT positions research delivery as a collaborative engagement rather than a static information transfer. Analysts work with clients to clarify objectives, interpret findings, and connect insights to strategic decisions.